Electrochemical (de)lithiation of silver ferrite and composites: mechanistic insights from ex situ, in situ, and operando X-ray techniques†
Abstract
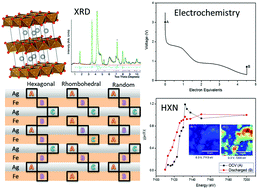
The structure of pristine AgFeO2 and phase makeup of Ag0.2FeO1.6 (a one-pot composite comprised of nanocrystalline stoichiometric AgFeO2 and amorphous γ-Fe2O3 phases) was investigated using synchrotron X-ray diffraction. A new stacking-fault model was proposed for AgFeO2 powder synthesized using the co-precipitation method. The lithiation/de-lithiation mechanisms of silver ferrite, AgFeO2 and Ag0.2FeO1.6 were investigated using ex situ, in situ, and operando characterization techniques. An amorphous γ-Fe2O3 component in the Ag0.2FeO1.6 sample is quantified. Operando XRD of electrochemically reduced AgFeO2 and Ag0.2FeO1.6 composites demonstrated differences in the structural evolution of the nanocrystalline AgFeO2 component. As complimentary techniques to XRD, ex situ X-ray Absorption Spectroscopy (XAS) provided insight into the short-range structure of the (de)lithiated nanocrystalline electrodes, and a novel in situ high energy X-ray fluorescence nanoprobe (HXN) mapping measurement was applied to spatially resolve the progression of discharge. Based on the results, a redox mechanism is proposed where the full reduction of Ag+ to Ag0 and partial reduction of Fe3+ to Fe2+ occur on reduction to 1.0 V, resulting in a Li1+yFeIIIFeIIyO2 phase. The Li1+yFeIIIFeIIyO2 phase can then reversibly cycle between Fe3+ and Fe2+ oxidation states, permitting good capacity retention over 50 cycles. In the Ag0.2FeO1.6 composite, a substantial amorphous γ-Fe2O3 component is observed which discharges to rock salt LiFe2O3 and Fe0 metal phase in the 3.5–1.0 V voltage range (in parallel with the AgFeO2 mechanism), and reversibly reoxidizes to a nanocrystalline iron oxide phase.


 Please wait while we load your content...
Please wait while we load your content...