Non-covalent interactions in anisole–(CO2)n (n = 1, 2) complexes
Abstract
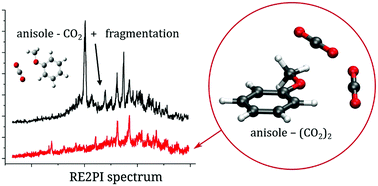
Non-covalent interactions are ubiquitous and represent a very important binding motif. The direct experimental measurement of binding energies in complexes has been elusive for a long time despite its importance, for instance, for understanding and predicting the structure of bio-macromolecules. Here, we report a combined experimental and computational analysis on the 1 : 1 and 1 : 2 clusters formed by anisole (methoxybenzene) and carbon dioxide molecules. We have obtained a detailed description of the interaction between CO2 and anisole. This system represents quite a challenging test for the presently available experimental and theoretical methods for the characterization of weakly bound molecular complexes. The results, evaluated in the framework of previous studies on anisole clusters, show a very good agreement between experimental and theoretical data. A comparison of the experimental and computational data enabled the binding energy values of the 1 : 1 and 1 : 2 clusters to be determined in the ground electronic state of the neutral and cation complex and in the first excited singlet state of the neutral complex. In addition, it was possible to adduce the presence of different 1 : 1+ conformers, prepared by direct ionization of the 1 : 1 complex or by dissociative ionization of the 1 : 2 complex.
- This article is part of the themed collections: 1st International Conference on Noncovalent Interactions and 2017 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...