Electrochemical CO2 reduction on Au surfaces: mechanistic aspects regarding the formation of major and minor products†
Abstract
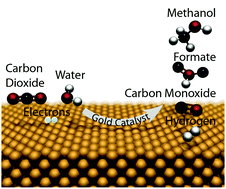
In the future, industrial CO2 electroreduction using renewable energy sources could be a sustainable means to convert CO2 and water into commodity chemicals at room temperature and atmospheric pressure. This study focuses on the electrocatalytic reduction of CO2 on polycrystalline Au surfaces, which have high activity and selectivity for CO evolution. We explore the catalytic behavior of polycrystalline Au surfaces by coupling potentiostatic CO2 electrolysis experiments in an aqueous bicarbonate solution with high sensitivity product detection methods. We observed the production of methanol, in addition to detecting the known products of CO2 electroreduction on Au: CO, H2 and formate. We suggest a mechanism that explains Au's evolution of methanol. Specifically, the Au surface does not favor C–O scission, and thus is more selective towards methanol than methane. These insights could aid in the design of electrocatalysts that are selective for CO2 electroreduction to oxygenates over hydrocarbons.


 Please wait while we load your content...
Please wait while we load your content...