Transition metal (Co, Ni) nanoparticles wrapped with carbon and their superior catalytic activities for the reversible hydrogen storage of magnesium hydride†
Abstract
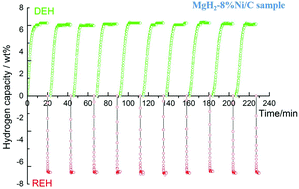
Magnesium hydride (MgH2) exhibits long-term stability and has recently been developed as a safe alternative to store hydrogen in the solid state, due to its high capacity of 7.6 wt% H2 and low cost compared to other metal hydrides. However, the high activation energy and poor kinetics of MgH2 lead to inadequate hydrogen storage properties, resulting in low energy efficiency. Nano-catalysis is deemed to be the most effective strategy in improving the kinetics performance of hydrogen storage materials. In this work, robust and efficient architectures of carbon-wrapped transition metal (Co/C, Ni/C) nanoparticles (8–16 nm) were prepared and used as catalysts in the MgH2 system via ball milling to improve its de/rehydrogenation kinetics. Between the two kinds of nano-catalysts, the Ni/C nanoparticles exhibit a better catalytic efficiency. MgH2 doped with 6% Ni/C (MgH2-6%Ni/C) exhibits a peak dehydrogenation temperature of 275.7 °C, which is 142.7, 54.2 and 32.5 °C lower than that of commercial MgH2, milled MgH2 and MgH2 doped with 6% Co/C (MgH2-6%Co/C), respectively. MgH2 doped with 6% Ni/C can release about 6.1 wt% H2 at 250 °C. More importantly, the dehydrogenated MgH2-6%Ni/C is even able to uptake 5.0 wt% H2 at 100 °C within 20 s. Moreover, a cycling test of MgH2 doped with 8% Ni/C demonstrates its excellent hydrogen absorption/desorption stability with respect to both capacity (up to 6.5 wt%) and kinetics (within 8 min at 275 °C for dehydrogenation and within 10 s at 200 °C for rehydrogenation). Mechanistic research reveals that the in situ formed Mg2Ni and Mg2NiH4 nanoparticles can be regarded as advanced catalytically active species in the MgH2-Ni/C system. Meanwhile, the carbon attached around the surface of transition metal nanoparticles can successfully inhibit the aggregation of the catalysts and achieve the steadily, prompting de/rehydrogenation during the subsequent cycling process. The intrinsic catalytic effects and the uniform distributions of Mg2Ni and Mg2NiH4 result in a favorable catalytic efficiency and cycling stability. Nano-catalysts with this kind of morphology can also be applied to other metal hydrides to improve their kinetics performance and cycling stability.


 Please wait while we load your content...
Please wait while we load your content...