ESR study of atomic hydrogen and tritium in solid T2 and T2:H2 matrices below 1 K†
Abstract
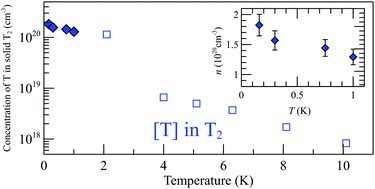
We report on the first ESR study of atomic hydrogen and tritium stabilized in solid T2 and T2:H2 matrices down to 70 mK. The concentrations of T atoms in pure T2 approached 2 × 1020 cm−3 (0.60%) and record-high concentrations of H atoms ∼1 × 1020 cm−3 (0.33%) were reached in T2:H2 solid mixtures where a fraction of T atoms became converted into H due to the isotopic exchange reaction T + H2 → TH + H. The maximum concentrations of unpaired T and H atoms were limited by their recombination which becomes enhanced by efficient atomic diffusion due to the presence of a large number of vacancies and phonons generated in the matrices by β-particles. Recombination also appeared in an explosive manner, both being stimulated and spontaneously in thick films where sample cooling was insufficient. We suggest that the main mechanism for H and T migration is physical diffusion related to tunneling or hopping to vacant sites in contrast to tunneling chemical exchange reactions which govern diffusion of H and D atoms created in H2 and D2 matrices by other methods.


 Please wait while we load your content...
Please wait while we load your content...