Reactions in silicon–nitrogen plasma†
Abstract
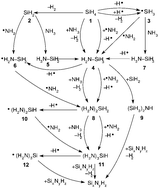
Reaction mechanisms that lead to creation of silicon–nitrogen bonds are studied in detail. These reactions are of fundamental importance for silicon nitride synthesis by plasma enhanced chemical vapour deposition from the gas mixture of silane (SiH4) and ammonia (NH3). All reactions in SiH4–NH3 plasma can be categorised as some of the basic types of reactions: bond dissociation, neutral nucleophilic substitution, radical neutralisation, neutral–radical addition, silylene addition, silylene rearrangement, radical nucleophilic addition or hydrogen abstraction reaction. Energetics of these reactions is analysed in detail for a great number of reactions belonging to these categories, by using theoretical modelling. Geometry optimisations are carried out with the MP2/aug-cc-pVTZ level of theory and energetics is further determined with high level ab initio calculations at the CASPT2/aug-cc-pVTZ level, which enabled confirmation of relevance of several mechanisms as reactions that lead to silicon nitride growth from plasma enhanced chemical vapour deposition, as well as introduction of new, energetically favourable mechanisms. Besides amine radical assisted eliminative addition and proton transfer reactions, silylene addition reactions are thermodynamically and kinetically favourable since they lack energy barriers. A new reaction pathway for synthesis of silicon nitride from plasma is proposed. This pathway is enabled by the ability of silylene to create two weak dative bonds, which enables silylene–amine complexes to stick to the silicon nitride surface. Upon dissociation of amine from the surface-bound complex, silylene remains on the surface, available for reaction with other reactive species from plasma.


 Please wait while we load your content...
Please wait while we load your content...