pH-regulated antimony oxychloride nanoparticle formation on titanium oxide nanostructures: a photocatalytically active heterojunction†
Abstract
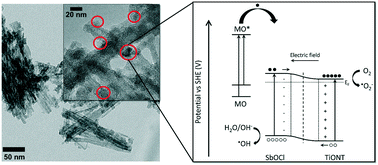
Improving the catalytic activity of heterogeneous photocatalysts has become a hot topic recently. To this end, considerable progress has been made in the efficient separation of photogenerated charge carriers by e.g. the realization of heterojunction photocatalysts. V–VI–VII compound semiconductors, namely, bismuth oxyhalides, are popular photocatalysts. However, results on antimony oxyhalides [SbxOyXz (X = Br, Cl, I)], the very promising alternatives to the well-known BixOyXz photomodifiers, are scarce. Here, we report the successful decoration of titanium oxide nanostructures with 8–11 nm diameter SbxOyXz nanoparticles for the first time ever. The product size and stoichiometry could be controlled by the pH of the reactant mixture, while subsequent calcination could transform the structure of the titanate nanotube (TiONT) support and the prepared antimony oxychloride particles. In contrast to the ease of composite formation in the SbxOyXz/TiONT case, anatase TiO2 could not facilitate the formation of antimony oxychloride nanoparticles on its surface. The titanate nanotube-based composites showed activity in a generally accepted quasi-standard photocatalytic test reaction (methyl orange dye decolorization). We found that the SbxOyClz/TiONT synthesized at pH = 1 is the most active sample in a broad temperature range.


 Please wait while we load your content...
Please wait while we load your content...