α-Diazo oxime ethers for N-heterocycle synthesis
Abstract
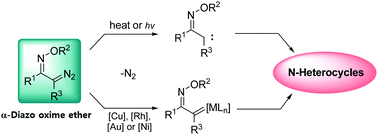
This Feature Article introduces the preparation and synthetic utility of α-diazo oxime ethers. α-Oximino carbenes are useful synthons for N-heterocycles, and can be easily prepared from α-diazo oxime ethers as precursors. We begin with the preparation of α-diazo oxime ethers and their application in [3+2] cycloaddition. It turns out that the nature of metals bound to carbenes plays a crucial role in modulating the reactivity of α-oximino carbenes, in which copper carbenes smoothly react with enamines, whereas the less reactive enol ethers and nitriles require gold carbenes. In Section 3.2, a discussion on N–O and C–H bond activation is presented. Carbenes derived from diazo oxime ethers show unique reactivity towards N–O and C–H bond activation, in which the proximity of the two functionalities, carbene and oxime ether, dictates the preferred reaction pathways toward pyridines, pyrroles, and 2H-azirines. In Section 3.3, the development of tandem reactions based on α-diazo oxime ethers is discussed. The nature of carbenes in which whether free carbenes or metal complexes are involved dissects the pathway and forms different types of 2H-azirines. The 2H-azirine formation turned out to be an excellent platform for the tandem synthesis of N-heterocycles including pyrroles and pyridines. In the last section, we describe the electrophilic activation of 2H-azirines with vinyl carbenes and oximino carbenes. The resulting azirinium species undergo rapid ring expansion rearrangements to form pyridines and pyrazines.


 Please wait while we load your content...
Please wait while we load your content...