Development of an enzyme cycling method by a purine nucleoside phosphorylase for assaying inorganic phosphate†
Abstract
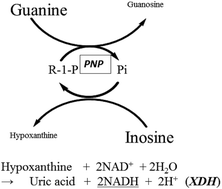
We have developed a novel enzymatic cycling method that uses purine nucleoside phosphorylase (PNP) (EC 2.4.2.1) from Bacillus sp. to measure inorganic phosphate. The method utilizes the reversibility of the PNP reaction, in which the forward and reverse reactions are catalyzed in the presence of an excess amount of inosine and guanine, respectively, a principle similar to that previously demonstrated with creatine kinase (CK). Real-time detection was accomplished by coupling the reaction with commercially available xanthine dehydrogenase (EC 1.17.1.4) in the presence of NAD+. The efficiency of the cycling reaction per unit of enzyme (U ml−1) was remarkably higher than the CK.


 Please wait while we load your content...
Please wait while we load your content...