Screening and identification of natural ligands of tyrosinase from Pueraria lobata Ohwi by a combination of ultrafiltration and LC-MS†
Abstract
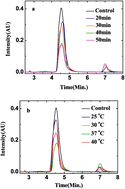
Ultrafiltration combined with liquid chromatography-mass spectrometry (UF-LC-MS) is an efficient affinity-based method to screen potential ligands of biomacromolecules from natural sources. In order to investigate the effects of constituents from Pueraria labta Ohwi on tyrosinase, a UF-LC-MS method for screening tyrosinase ligands was established. Seven constituents were screened as tyrosinase ligands with high affinity and their structures were elucidated as isoflavones and isoflavone glycosides by mass spectrum analysis, among which puerarin exhibited much stronger inhibitory activity in vitro than arbutin against tyrosinase. An in silico docking study indicates that the interaction between puerarin and tyrosinase mainly derived from hydrogen bonds and π–cation interaction.


 Please wait while we load your content...
Please wait while we load your content...