A pyridine based meta-linking deep-blue emitter with high conjugation extent and electroluminescence efficiencies†
Abstract
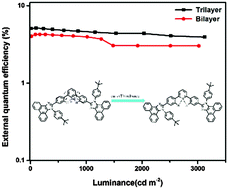
We designed and synthesized a bipolar deep-blue emitter 2,6-bis(4-(1-(4-(tert-butyl)phenyl)-1H-phenanthro[9,10-d]imidazol-2-yl)phenyl)pyridine (26BTPIPy) based on a meta-linking D–π–A–π–D structure. Compared to its para-linking analogue (25BTPIPy), the meta-linking in 26BTPIPy effectively shortens molecular conjugated length and restricts intramolecular charge transfer. Interestingly, unlike most other meta-linking emitters, a high fluorescence yield can be maintained in 26BTPIPy. This may be attributed to a relatively planar structure at the benzene–pyridine–benzene joint in 26BTPIPy leading to considerable overlapping of its frontier molecular orbitals. These suitable combinations of properties endow 26BTPIPy with efficient deep-blue emission and good bipolar carrier transporting characteristics. An organic light-emitting device using 26BTPIPy as an emitter shows a low turn-on voltage (2.8 V), deep-blue emission with a color index of (0.15, 0.09) and high current and external quantum efficiencies (4.16 cd A−1 and 5.15%). Besides, a bilayer device using 26BTPIPy as both an emitting and electron-transporting material also gives high performance with a current efficiency of 4.22 cd A−1 and a color purity of (0.15, 0.11).

 Please wait while we load your content...
Please wait while we load your content...