Bis(2-oxoindolin-3-ylidene)-benzodifuran-dione and bithiophene-based conjugated polymers for high performance ambipolar organic thin-film transistors: the impact of substitution positions on bithiophene units†
Abstract
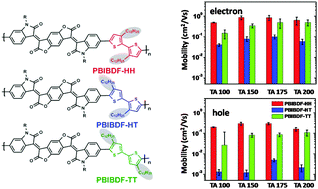
Three donor–acceptor polymers based on (3E,7E)-3,7-bis(2-oxoindolin-3-ylidene)benzo-[1,2-b:4,5-b]-difuran-2,6(3H,7H)-dione (BIBDF) and three kinds of dialkylated bithiophenes with head-to-head (HH), head-to-tail (HT), and tail-to-tail (TT) connectivity were synthesized by the Stille coupling reaction. Their photophysical and electrochemical properties, electronic device performance, and microstructure were investigated. We found that the alkyl chains substituted near the thiophene–thiophene connection cause less steric hindrance than those near the BIBDF–thiophene connection. Therefore PBIBDF-HH exhibited the preferred planarity, crystallinity, and molecular orientation, yielding the highest field-effect mobility. A maximum electron mobility of 1.23 cm2 V−1 s−1 and a maximum hole mobility of 0.37 cm2 V−1 s−1 were obtained for PBIBDF-HH-based devices. These results show that the substitution sites on the bithiophene units play an important role in the determination of molecular organization and the resulting device performance.

 Please wait while we load your content...
Please wait while we load your content...