Supramolecular hydrogelation with bile acid derivatives: structures, properties and applications
Abstract
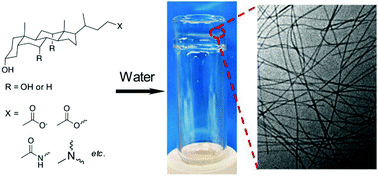
Hydrogelation of small molecules in aqueous solutions results from a balance between solubilization and precipitation (or crystallization). The hydrophobic moieties of amphiphiles tend to aggregate and the hydrophilic units may stabilize the aggregates in aqueous solutions. Morphologies vary according to the chemical structure of the amphiphiles. The formation of nanofibers or worm-like micelles is a prerequisite for hydrogels. Molecular hydrogels often show better degradability and functional diversity than polymeric hydrogels and may be useful in biomedical applications. Bile acids have attracted increasing attention for designing various biomaterials, including molecular hydrogels. They are naturally occurring amphiphilic compounds that exist in our body and help with the dissolution and digestion of fat by the formation of micelles. This review highlights the recent progress in the field of molecular hydrogelators based on bile acids, including bile salts, anionic, cationic and neutral bile acid derivatives, two-component hydrogelation systems, and polymeric supramolecular hydrogels, along with their potential applications.

 Please wait while we load your content...
Please wait while we load your content...