Dual-valence nickel nanosheets covered with thin carbon as bifunctional electrocatalysts for full water splitting†
Abstract
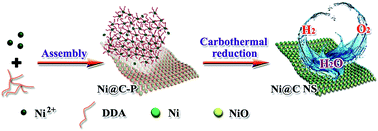
The viability of clean hydrogen energy strongly depends on the efficient non-precious metal-based electrocatalysts. Herein, thin carbon coated nickel-based nanosheets (Ni@C NSs) were designed and synthesized by controlling the successive assembly and carbothermal reduction under an inert atmosphere. The nanosheets were assembled by partially reduced Ni nanoparticles and NiO nanoparticles wrapped in thin graphitic carbon. Dodecyl amine played a vital role in the structure-directing of the interconnected nanosheets by suppressing the nanoparticles growth and providing a protective effect on the low-valence nickel. The resulting metallic Ni and NiO were the effective active sites for hydrogen and oxygen evolution, respectively; moreover, the neighboring other nickel species could exert a good synergetic effect on the electrocatalytic reaction. The outside thin carbon film was also conducive to the electrochemical stability. Furthermore, based on the practical application considerations, the Ni@C NSs were directly grown on the Ni foam surface. The water electrolyser made up of Ni@C NSs/Ni foam electrodes presented a 1.57 V cell voltage in an alkaline medium accompanied with long-term durability. The highly active transition-metal catalysts with two valences is promising for full water splitting devices.

 Please wait while we load your content...
Please wait while we load your content...