Poly(ionic liquid) hydrogels exhibiting superior mechanical and electrochemical properties as flexible electrolytes†
Abstract
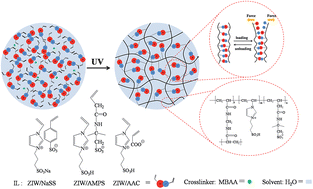
Recently, flexible electrolytes have aroused tremendous interest due to their wide applications in flexible electronic devices, such as wearable electronics, roll-up displays, smart mobile devices and implantable biosensors. Herein, novel ionic liquids, whose both cations and anions both can be polymerized, are used to construct flexible poly(ionic liquid) hydrogel electrolytes by one-step synthesis. Originating from the ionic interactions inside the ionic liquids, these hydrogels combine strong and weak crosslinks after polymerization, which makes these hydrogels tough, stretchable, flexible and self-recoverable. Their superior mechanical properties have been certified by mechanical tests. Moreover, these poly(ionic liquid) hydrogels show extremely high ionic conductivities over 1 S m−1 at room temperature and low activation energy. Notably, their electrochemical behaviors can remain stable under different bending angles and successive bending, folding, compressing and twisting. As flexible electrolytes, these poly(ionic liquid) hydrogels give the possibility to be used in fuel cells or supercapacitors, thus, promoting the development of flexible electronic devices.

 Please wait while we load your content...
Please wait while we load your content...