Facile growth of porous Fe2V4O13 films for photoelectrochemical water oxidation†
Abstract
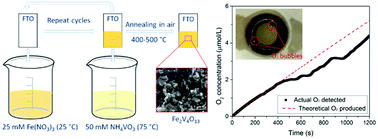
Porous n-type Fe2V4O13 films on FTO substrates were prepared by a simplified successive ion layer adsorption and reaction method and characterized as photoelectrodes for photoelectrochemical (PEC) water oxidation. Synthesis parameters such as film thickness and annealing temperatures and durations were investigated to optimize the PEC performance. A band gap of ∼2.3 eV and a flat band potential of 0.5 V vs. RHE make Fe2V4O13 a promising photoanode material. Water oxidation was kinetically limited at the surface of Fe2V4O13 film as confirmed by tests in electrolyte with a hole scavenger (Na2SO3). Improved PEC performance was achieved by Mo and W doping because of enhanced carrier densities. The best performance was obtained by 2.5% W-doped Fe2V4O13 films (actual 0.8% W-doped), which efficiently oxidize water to O2via photogenerated holes as confirmed by oxygen evolution measurements. Moreover, the Fe2V4O13 photoanode displayed very stable photocurrent under illumination. Due to the suitable band gap and valence band position, Fe2V4O13 is a promising photoanode for solar water splitting. Co-catalyst loading and doping optimization are identified as routes to improve this material's performance further.
- This article is part of the themed collection: Water splitting and photocatalysis

 Please wait while we load your content...
Please wait while we load your content...