Intramolecular multi-bond strain: the unrecognized side of the dichotomy of conjugated systems
Abstract
Electron conjugation stabilizes unsaturated systems and diminishes the differences among bond distances. Experimentally, Kistiakowsky and coworkers first measured and noticed the difference between the hydrogenation heats of carbon–carbon double bonds in conjugated systems. For instance, the hydrogenation heat of butadiene is 57.1 kcal mol−1, which is less than two times that of the hydrogenation heat of 1-butene (30.3 kcal mol−1), and the difference (3.5 kcal mol−1) is the extra stabilization due to the resonance between two double bonds in the former, and is referred to as the experimental resonance energy. Following Kistiakowsky's definition, Rogers et al. studied the stepwise hydrogenation of 1,3-butadiyne and concluded that there is no conjugation stabilization in this molecule. This claim received objections instantly, but Rogers and coworkers further showed the destabilizing conjugation in 2,3-butanedione and cyanogen. Within resonance theory, the conjugation energy is derived “by subtracting the actual energy of the molecule in question from that of the most stable contributing structure.” The notable difference between the experimental and theoretical resonance energies lies in that the former needs other real reference molecules while the latter does not. Here we propose and validate a new concept, intramolecular multi-bond strain, which refers to the repulsion among π bonds. The π–π repulsion, which is contributed to by both Pauli exchange and electrostatic interaction, is quantified with the B4H2 model system (16.9 kcal mol−1), and is compared with the σ–σ repulsion in B2H4 (7.7 kcal mol−1). The significance of the π–π repulsion can be demonstrated by the much longer carbon–nitrogen bond in nitrobenzene (1.486 Å) than in aniline (1.407 Å), the very long and weak nitrogen–nitrogen bond (1.756 Å) in dinitrogen tetroxide, and the instability of long polyynes. This new concept successfully reconciles the discrepancy between experimental and theoretical conjugation energies. However, we maintain that by definition, electron conjugation must be stabilizing.
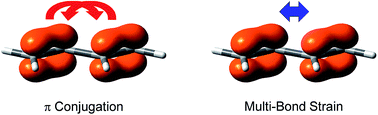
- This article is part of the themed collection: Celebrating a Century of Excellency in Chemistry at Xiamen University

 Please wait while we load your content...
Please wait while we load your content...