Diene hydroaminomethylation via ruthenium-catalyzed C–C bond forming transfer hydrogenation: beyond carbonylation†
Abstract
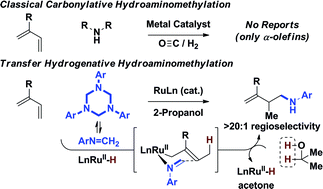
Under the conditions of ruthenium catalyzed transfer hydrogenation using 2-propanol as terminal reductant, 1,3-dienes engage in reductive C–C coupling with formaldimines obtained in situ from 1,3,5-tris(aryl)-hexahydro-1,3,5-triazines to form homoallylic amines. Deuterium labelling studies corroborate a mechanism involving reversible diene hydroruthenation to form an allylruthenium complex that engages in turn-over limiting imine addition. Protonolysis of the resulting amidoruthenium species releases product and delivers a ruthenium alkoxide, which upon β-hydride elimination closes the catalytic cycle. These transformations, which include enantioselective variants, represent the first examples of diene hydroaminomethylation.

 Please wait while we load your content...
Please wait while we load your content...