Inter-moieties reactivity correlations: an approach to estimate the reactivity endpoints of major atmospheric reactants towards organic chemicals†
Abstract
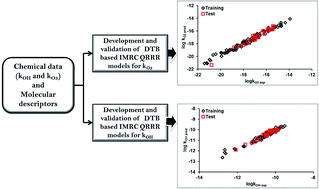
The hydroxyl (OH) radical and ozone (O3) are two of the major reactive species that largely determine the persistence of atmospheric chemicals. Developing experimental protocols for determining their reaction rate kinetics with an enormously large number of chemicals would be difficult and time and cost intensive; however, computational approaches offer the possibility to overcome such problems. Inter-moieties reactivity correlations (IMRC) provide a tool for developing quantitative reactivity–reactivity relationship (QRRR) models for predicting the kinetic rate constants for the atmospheric reactions of the OH radical and O3 with chemicals. In the present study, we propose the IMRC QRRR models for estimation of the rate constants for the OH radical and O3 reactions with atmospheric chemicals. The rigorously validated models in test data yielded correlations (R2) of >0.94 between the experimental and predicted reactivity endpoints and performed better than earlier reported quantitative structure–reactivity relationship models. The developed QRRR models were also able to offer mechanisms for the OH radical and O3 reactions with atmospheric chemicals. The applicability domains of the developed models were defined using the leverage and standardization methods. The proposed models can be employed for extrapolating reactivity endpoints to OH radical and O3 reactions with chemicals when the data for the other reactant are available.

 Please wait while we load your content...
Please wait while we load your content...