Metal–organic frameworks MIL-88A with suitable synthesis conditions and optimal dosage for effective catalytic degradation of Orange G through persulfate activation
Abstract
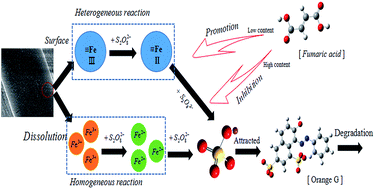
MIL-88A was synthesized in diverse preparation conditions and characterized by various techniques. The catalytic performance of MIL-88A series for degrading Orange gelb (OG) through persulfate activation were also tested at temperature of 25 °C and the results were shown in the following sequence: 85 °C/2 h > 65 °C/2 h > 105 °C/2 h > 65 °C/6 h > 65 °C/12 h. MIL-88A produced at the synthesis temperature of 85 °C and in the crystallization time of 2 h (85 °C/2 h) was the best one with a removal rate of 96.4% mainly due to high SBET and much greater leach-out of Fe. An optimal dosage (0.3 g L−1) for MIL-88A in the MIL-88A/PS/OG system exists because of inhibition and promotion by fumaric acid. The filtrate experiment indicated that the catalytic activation involved heterogeneous reactions on the surface of the catalyst and homogeneous reactions in solutions. But the heterogeneous reaction occupied the principal position and the probable mechanism was obtained. Moreover, only solution pH < 4 could show a high degradation effect without the help of temperature. Comparing two different methods in the recycling experiment, the removal rate of OG decreased after the fourth run in both. Loss of active catalytic sites for Fe(III) in MIL-88A in the process of separating and sampling was responsible for activity decay.

 Please wait while we load your content...
Please wait while we load your content...