Spectroscopic study on spontaneously grown silver@ultra-thin cerium oxide nanostructures
Abstract
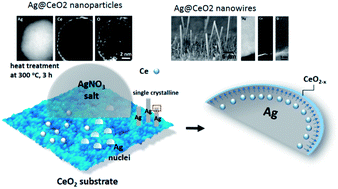
Ag@CeO2 nanostructures have been recently reported to show unique catalytic properties but synthetic methods for them are limited. This study investigates microstructural characteristics of Ag@CeO2 nanowires and nanoparticles with a spontaneously-grown ultra-thin ceria shell (∼0.5 nm), which were synthesized on CeO2 substrate without using oxide precursors. Elemental mapping and line scanning by electron energy loss spectroscopy (EELS) suggest that the Ce in the CeO2 substrate dissolved in the molten silver nitrate salt and was repelled to the surface of the Ag nanostructures to form continuous oxide shells during the growth of Ag single-crystals. X-ray absorption near edge structure (XANES) spectra verify that the valence of the Ce ions in the oxide layer was between Ce3+ and Ce4+.


 Please wait while we load your content...
Please wait while we load your content...