Monofunctional platinum(ii) dithiocarbamate complexes: synthesis, characterization and anticancer activity†
Abstract
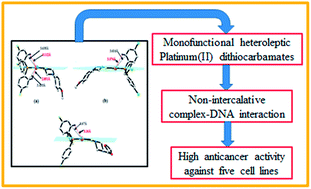
Heteroleptic platinum(II) dithiocarbamates, of general formula [Pt(DTC)LCl], where DTC = 4-(4-methoxyphenyl)piperazine-1-carbodithioate (1 and 2) and 4-(furan-2-carbonyl)piperazine-1-carbodithioate (3) and L = tri(4-flourophenylphosphine) (1 and 3) and tri(4-chlorophenylphosphine) (2) have been synthesized and characterized by different analytical techniques. These complexes are square planar with picoplatin or phenanthriplatin type steric hindrance from aromatic C–H groups of the phosphine ligand as shown by single-crystal analysis. In 1, the Pt square plane is hindered by two axially oriented hydrogens, whereas by only one in 2 and 3. DNA-binding studies by UV/visible spectroscopy revealed a stronger electrostatic interaction of 1 compared to 2 and 3, and the results are further supported by viscometry and cyclic voltammetric measurements. Their in vitro anticancer activity against five different cancer cell lines using a MTT assay revealed high potency of the complexes. The higher activity of 1 than both 2 and 3 is consistent with DNA binding strength and we speculate that it may be due to the relatively inert nature of platinum towards off-target biomolecules ensured by the hindrance caused by the two axially oriented hydrogens.


 Please wait while we load your content...
Please wait while we load your content...