A porous magnesium metal–organic framework showing selective adsorption and separation of nitrile guest molecules†
Abstract
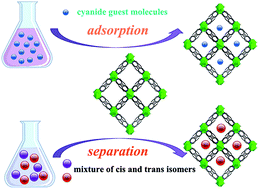
The porous metal–organic framework (MOF) {[Mg2(BDC)2(BPNO)]·2DMF (Mg-MOF, mother crystal; DMF = N,N-dimethylformamide) was prepared by the solvothermal reaction of 1,4-benzene dicarboxylate (H2BDC), 4,4′-dipyridyl-N,N′-dioxide (BPNO) and Mg(NO3)·6H2O in DMF/EtOH. This Mg-MOF has a unique structure with 1-dimensional (1D) channel as well as infinite metal chains, which resembles the topology of MIL-53. Desolvated Mg-MOF can be used as absorbent for selective adsorption and separation of nitrile guest molecules including acetonitrile (MeCN), acrylonitrile (ACN), allylnitrile (AN) and crotononitrile (CTN) without loss of crystallinity. Furthermore, desolvated Mg-MOF also selectively captured cis-crotononitrile into its cavity from a mixture of cis and trans isomers. The process of adsorption produced quantitatively four daughter crystals, [Mg2(BDC)2(BPNO)]·2S (2: 2A, S = MeCN; 2B, S = ACN; 2C, S = AN; 2D, S = cis-CTN), which exhibit reversible interconversion by guest-exchanges at RT in single crystal transformations. Vapor phase adsorption isotherms of MeCN, ACN, AN and CTN on the desolvated Mg-MOF are presented.

 Please wait while we load your content...
Please wait while we load your content...