Chitosan and imide-functional Fe3O4 nanoparticles to prepare new xanthene based poly(ether-imide) nanocomposites
Abstract
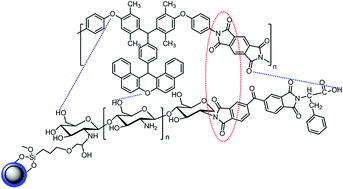
In this work a new series of multifunctional nanocomposites were synthesized based on a soluble poly(ether-imide). The poly(ether-imide) was successfully synthesized via direct polycondensation reaction. A methyl rich diamine containing xanthene group as a new monomer was synthesized and used for polymerization. The xanthene based poly(ether-imide) was characterized using size exclusion chromatography (SEC) and nuclear magnetic resonance (NMR). Fe3O4 nanoparticles were imide-functionalized by a dianhydride, chitosan and phenylalanine. The functional Fe3O4 nanoparticles were incorporated into the new synthesized poly(ether-imide). Effects of the functionalized Fe3O4 nanoparticles on the thermal and combustion properties of the corresponding nanocomposites were investigated. Thermogravimetric analysis (TGA) results indicated that the thermal stability and char yields of the nanocomposites were enhanced compared to the neat poly(ether-imide). Microscale combustion calorimetry (MCC) revealed that the synthesized poly(ether-imide) had low flammability. The incorporation of functional Fe3O4 nanoparticles could further improve the combustion properties of poly(ether-imide). The high interaction between poly(ether-imide) and functional Fe3O4, the presence of an imide group and high hydroxyl content of the functional Fe3O4 nanoparticles seem to be responsible for the improvement of the thermal and combustion properties. Furthermore, the presence of methyl, ether and bulky xanthene groups in the polyimide backbone decreased the glass transition temperature (Tg) and increased the solubility in organic solvents. These properties will be useful for processing and new applications of poly(ether-imide).


 Please wait while we load your content...
Please wait while we load your content...