Anion assisted supramolecular self-assemblies of succinate and malate adducts: crystal structures and theoretical modelling†
Abstract
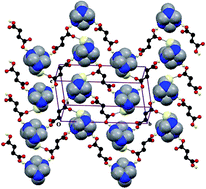
A series of five dicarboxylic acid complexes of succinic acid and malic acid, hexamethylenetetramine succinic acid (HMTSA) (1), bis(3-methylanilinium) succinate succinic acid (3MASSA) (2), 4-methoxyanilinium hydrogen succinate (4MAHS) (3), 2-aminoanilinium hydrogen malate (2AAHM) (4) and 4-ethoxyanilinium hydrogen malate (4EAHM) (5), have been synthesized and structurally analyzed; also, the supramolecular organizations within their crystalline solids have been studied. Strong hydrogen bonds such as O–H⋯O, N–H⋯O and O–H⋯N are dominant in the formation of one- and two-dimensional supramolecular frameworks. In addition, C–H⋯O and C–H⋯N weak interactions also play important roles in the framework of supramolecular self-assembly. The geometries of the crystal structures have been optimized by the DFT/B3LYP method using the 6-31g(d,p) basis set, and the results have been found to be comparable with experimental results. Computational analysis of the crystal packing by the PIXEL method provided deeper insight into the nature and domination of intermolecular interactions in the overall stabilization. Hirshfeld surfaces and their associated 2D fingerprint plot analyses revealed that closer molecular contacts may aid the crystal packing. The reported compounds were also characterized with FT IR spectroscopy and thermogravimetric analysis.

 Please wait while we load your content...
Please wait while we load your content...