Theoretical insight into the Au(i)-catalyzed hydration of halo-substituted propargyl acetate: dynamic water-assisted mechanism†
Abstract
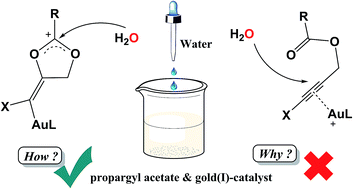
The hydration mechanism of halo-substituted propargyl acetate, catalyzed by a homogenous Au(I) complex, has been investigated with the aid of the density functional theory (DFT) method. Our results reveal that the hydration is initiated by the favoured 1,5-exo-dig cycloaddition in the anti manner, affording a desired regioselective Markovnikov product. We also verify that neither the pathway towards the anti-Markovnikov product triggered by 1,6-endo-dig cycloaddition, nor direct nucleophilic attack by water, would happen without the help of neighbouring carbonyl groups. The favoured pathway mainly includes three processes: nucleophilic attack after 1,5-exo-dig cycloaddition, protodeauration, and enol–keto tautomerization. It turns out that the third process (enol–keto tautomerization) is the rate-determining step. Additionally, different halo-substituents cannot change the reaction trend, but slightly affect the relative energies. Particularly, cluster-continuum solvent models were established for some proton-transfer steps to rationally simulate reaction processes and evaluate energy barriers. Our study suggests that the presence of an explicit water-bridge is crucial to promote the hydration reaction. Computational results provide theoretical support for experimental observations, and insight into the hydration.

 Please wait while we load your content...
Please wait while we load your content...