Determining the substrate permeability through the bilayer of large unilamellar vesicles of DOPC. A kinetic study
Abstract
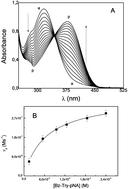
In this work we determine the permeability of 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine (DOPC) vesicles in the presence of different cholesterol (Cho) contents, by using the enzymatic hydrolysis of N-benzoyl-L-tyrosine p-nitroanilide (Bz-Try-pNA) catalyzed by α-chymotrypsin (α-CT). The reaction was first studied in homogeneous media in a 4% p/p ethanol–water mixture and then in DOPC vesicles at different Cho content. In homogenous media, ethanol helps to solubilize the substrate, which is almost insoluble in water and therefore increases the effective concentrations. In DOPC vesicles, ethanol does not destroy the bilayer. In both cases, the enzymatic hydrolysis can be followed by UV-visible spectroscopy. The hydrolysis of Bz-Try-pNA catalyzed by α-CT follows the Michaelis–Menten mechanism and the kinetic parameters: kcat, KM and kcat/KM were evaluated in both systems at the same solvent mixture compositions. To obtain the kinetic parameters and the permeability of the reactant in DOPC : Cho vesicles, we use a simple mathematical model and dynamic light scattering (DLS) measurements. The results show that the hydrolysis reaction takes place in the water entrapped in the interior of the DOPC vesicles and, that the enzyme encapsulated inside the vesicles, despite the significant differences in the permeability values of Bz-Try-pNA, has similar catalytic effects independently on the Cho composition used.

 Please wait while we load your content...
Please wait while we load your content...