Aqueous phase reforming and hydrodeoxygenation of ethylene glycol on Pt/SiO2–Al2O3: effects of surface acidity on product distribution†
Abstract
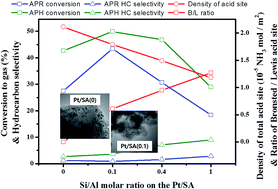
Aqueous-phase reforming (APR) and aqueous-phase hydrodeoxygenation (APH) reactions of ethylene glycol (EG) were investigated using platinum supported solid-acid SiO2–Al2O3 catalysts with different Si/Al molar ratios. The molar ratio of Si/Al on the SiO2–Al2O3 mixed metal oxides largely altered the surface area due to changes to the acidity, as well as changing the reduction behavior of the supported platinum nanoparticles. The Pt/SiO2–Al2O3 catalysts with a Si/Al molar ratio of 0.1 showed a higher activity for APR as well as APH. Among the various properties of Pt/SiO2–Al2O3, the amount of acid sites on the SiO2–Al2O3 supports changed the EG conversion and production distribution with different coke depositions. The acidic property was a more dominant factor for the catalytic activity than the affects of the platinum crystallite size on the reduction behavior. The easy and simultaneous cleavages of C–C as well as the C–O bonds in EG on the Bronsted acid sites of Pt/SiO2–Al2O3 catalysts were responsible for a higher EG conversion and hydrocarbon formation. A larger number of weak acid sites was also related to the formation of larger hydrocarbons and a lower coke deposition. Compared with Pt/Al2O3, improved catalytic acidity with a low coke deposition was observed for Pt/SiO2–Al2O3 at a Si/Al molar ratio of 0.1. This can mainly be attributed to the easy control of weak and strong acid sites with a high dispersion of platinum crystallites by simply changing the Si/Al molar ratio of the SiO2–Al2O3 mixed metal oxides.

 Please wait while we load your content...
Please wait while we load your content...