Site-preferential occupancy induced photoluminescence tuning in (Ca,Ba)5(PO4)3Cl:Eu2+ phosphors†
Abstract
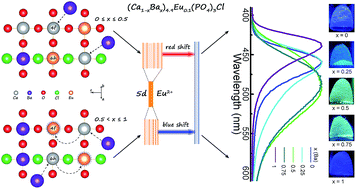
Apatite structured (Ca1−xBax)5(PO4)3Cl:Eu2+ (0 ≤ x ≤ 1) solid-solution phosphors were successfully prepared via a typical Pechini sol–gel method. Structural refinement confirms the formation of solid-solution phases under the whole substitution ratio. All samples crystallized in a hexagonal phase with a space group of P63/m (176), and there are two kinds of cation sites (4f and 6h) in the host lattice. The as-prepared (Ca1−xBax)5(PO4)3Cl:Eu2+ phosphors show similar broad absorptions from 250 nm to 450 nm with peaks around 397 nm. Under 397 nm UV, Ca5(PO4)3Cl:Eu2+ presents a blue emission centered at 460 nm. By substituting Ba2+ for Ca2+, an unexpected red-shift up to 490 nm was first observed at x ≤ 0.5 and then a blue-shift after x > 0.5 occurred with a resulting emission at 438 nm for x = 1. The abnormal red-shift and blue-shift were revealed by the Rietveld structural refinement method and the possible luminescence mechanisms were proposed. The former is attributed to the preferential occupancy of 6h sites by Eu2+ ions and the expansion of neighboring Ba/Ca–O bond lengths. The latter mainly results from the entering of Eu2+ ions into the looser Ba2+ sites. The proposed luminescence mechanism can help reveal the underlying mechanisms in optical adjustment by changing the coordination environment at local sites. In addition, the thermal stability of (Ca,Ba)5(PO4)3Cl:Eu2+ phosphors were systematically investigated. Generally, the as-prepared (Ca,Ba)5(PO4)3Cl:Eu2+ phosphors can act as potential emitting-tunable phosphors for potential applications in n-UV based white LEDs.

 Please wait while we load your content...
Please wait while we load your content...