Preparation of redox-sensitive, core-crosslinked micelles self-assembled from mPEGylated starch conjugates: remarkable extracellular stability and rapid intracellular drug release†
Abstract
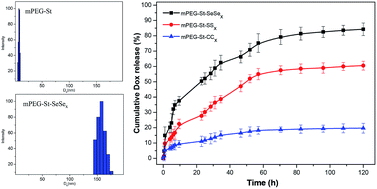
An ideal tumor drug delivery platform should offer little release of the drug in a physiological environment and fast release of the drug once endocytosis into the tumor tissues occurs. Therefore, we have developed a new kind of core-crosslinked copolymers (mPEG-St-SeSex) based on mPEGylated starch and diselenide bonds for rapid drug release. The corresponding core-crosslinked analogs mPEG-St-SSx and mPEG-St-CCx copolymers using 3,3′-dithiodipropionic acid and octanedioic acid as crosslinkers, respectively, were synthesized and used as controls. The results displayed that all amphiphilic core-crosslinked copolymers could form micelles with spherical structures in aqueous solution, showing advanced stability compared to non-crosslinked micelles (mPEG-St) in physiologically related and ultra micelle destabilization conditions. The disassembly of micelles triggered by DTT, combined with in vitro drug release assay and intracellular drug release studies using confocal laser scanning microscopy (CLSM), indicated that the DOX-loaded mPEG-St-SeSex micelles showed higher activity and could be more quickly disrupted than the DOX-loaded mPEG-St-SSx and mPEG-St-CCx micelles in reducing environment and subsequently rapidly released drug. These results, in addition to cell cytotoxicity studies, demonstrated that the mPEG-St-SeSex micelles have the potential to develop controlled drug-delivery systems for cancer therapy.

 Please wait while we load your content...
Please wait while we load your content...