Generation of hydrogen peroxide and hydroxyl radical resulting from oxygen-dependent oxidation of l-ascorbic acid via copper redox-catalyzed reactions
Abstract
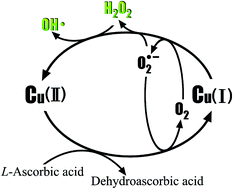
The generation of hydrogen peroxide (H2O2) and hydroxyl radical (HO˙) during the oxidation of L-ascorbic acid (L-AA) by oxygen with copper as a catalyst was investigated to set up the O2/Cu/L-AA process with benzoic acid (BA) as a probe reagent. The high concentration of H2O2 that is generated undergoes an intramolecular two-electron transfer and is further activated by the intermediate cuprous copper [Cu(I)] to yield HO˙ as a product, resulting in significant degradation of BA. Dehydroascorbic acid, 2,3-diketogulonic acid, and L-xylosone were the predominant detected products of the oxidation of L-AA. However, the generation of H2O2 and degradation of BA were regulated by variations in pH, which results from the contradiction between protonated L-AA that is difficult to chelate with Cu(II) via electron transfer and hydrogen ions (H+), which are indispensable for the generation of H2O2. Furthermore, the concentration of H2O2 and degradation of BA increased with an increase in the dosage of L-AA. Trace amounts of Cu(II) are effective for catalyzing the oxidation of L-AA, whereas the generation of H2O2 and degradation of BA increased with an increase in the dosage of Cu(II). Owing to the formation of Cu(I) chloride complexes or Cu(II) chloride complexes, the addition of chloride (Cl−) could inhibit the generation of H2O2 and degradation of BA.

 Please wait while we load your content...
Please wait while we load your content...