Fast flow synthesis of highly reactive polyisobutylene co-initiated by an AlCl3/isopropyl ether complex
Abstract
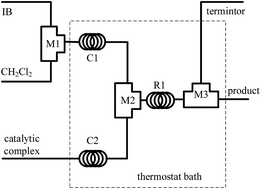
In this work, with AlCl3 addition in the range from 4 to 10 mmol L−1 and enough isopropyl ether (iPr2O) addition, we successfully synthesized highly reactive polyisobutylene (HRPIB) using a microflow system within 12 s or less. The temperature window was extended from −20 °C to 50 °C, and the molecular weight (Mn) was adjustable between 500 and 15 000. The evolutions of HRPIBs and the effects of reaction conditions were carefully investigated, revealing multiple effects of an excess of iPr2O over AlCl3 including: (1) decreasing the intensive isomerization co-initiated by free AlCl3; (2) inhibiting the chain termination to present chain transfer dominated kinetics; (3) weakening the temperature sensitivity of Mn as a restriction on temperature elevation; (4) retarding the chain propagation to slow the reaction and increase the probability of isomerization. Key access to fast synthesis of high quality HRPIB is proposed to make a compromise of these effects by properly selecting iPr2O : AlCl3.

 Please wait while we load your content...
Please wait while we load your content...