Microwave-assisted preparation of SO42− intercalated hydrotalcites for ammonia–nitrogen removal
Abstract
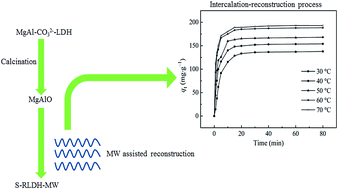
The intercalation of SO42− into MgAl hydrotalcites was investigated under microwave irradiation, and the structural and physiochemical properties of the SO42− intercalated hydrotalcites were determined using XRD, FTIR, SEM/EDS, BET and BJH techniques. Microwave irradiation contributed to the intercalation of SO42− anions, which brought about high amounts of SO42− into the interlayer spaces and high crystallinity. The intercalation of SO42− enhanced the acidic sites that cooperated with alkaline sites to activate the NH4+ molecules simultaneously leading to a significant increase in the removal of ammonia–nitrogen in synthetic wastewater. Kinetics and thermodynamics of the SO42− intercalation under microwave irradiation revealed that Langmuir isotherm fitted the experimental results well, and the kinetics were more accurately represented by a pseudo second-order model. The intercalation–reconstruction process was chemisorption nature mainly through hydrogen bond forces, followed the exchange of interlayer –OH anions and SO42−.

 Please wait while we load your content...
Please wait while we load your content...