Quorum quenching activity in the cell-free lysate of Enterobacter ludwigii isolated from beef and its effect on quorum sensing regulation in Yersinia enterocolitica†
Abstract
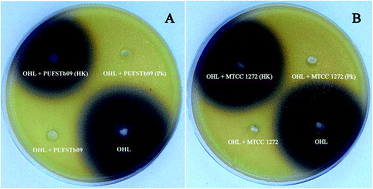
A wide range of Gram negative bacteria regulate their phenotypic characteristics through an auto-inducing signaling mechanism called Quorum Sensing (QS). The AHL-mediated quorum sensing mechanism plays a crucial role in biofilm formation and other virulence factors. Hence, disrupting the QS signaling mechanism might be an effective approach. In this study, bacterial isolates from beef were screened for their quenching activity. The cell-free lysates of test bacterial isolates were confirmed for their quenching activity using the reporter strain C. violaceum CV026 and quantified by means of HP-TLC. In silico techniques like molecular docking and dynamics simulation studies were applied to the predicted tertiary structure of lactonase. The cell-free lysate of Enterobacter ludwigii PUFSTb09 (KR476387) exhibited pronounced quorum quenching activity by reducing 78.20% of violacein production by the reporter strain. Furthermore, the cell-free lysate of test bacteria exhibited significant (p < 0.05) reduction in biofilm formation and EPS production of Y. enterocolitica (KT266804) by 66.15% and 70.18% respectively. The biofilm inhibitory activity of the test isolate was further confirmed by means of confocal laser microscopy and scanning electron microscopy, which revealed the suppression of total biomass. Molecular analysis revealed the presence of the aiiA gene in Enterobacter ludwigii PUFSTb09 and in docking analysis it was predicted that the affinity towards the OHL–lactonase complex was less than that of LasR–OHL complex, which might be due to the degrading nature of lactonase enzyme towards the natural ligand. The study shows the potential of AHL degradation by AHL lactonase present in the cell-free lysate of E. ludwigii, and regulation of QS-dependent phenotypes in food-borne Y. enterocolitica.

 Please wait while we load your content...
Please wait while we load your content...