Determination of hypochlorite by quenching the fluorescence of 1-pyrenylboronic acid in tap water†
Abstract
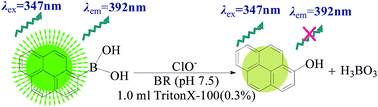
In neutral conditions, hypochlorite-assisted oxidative conversion of 1-pyrenylboronic acid into 1-hydroxypyrene, which leads to 1-pyrenylboronic acid fluorescence quenching, was used as the signaling tool. Compared with 1-pyrenylboronic acid, the maximum excitation (λex = 347 nm) and emission (λem = 392 nm) wavelength of 1-hydroxypyrene had no obvious change. The surfactant Triton X-100, as a micellar additive, was not only used to enhance the stability of the fluorescence probe, but also to improve its sensitivity. When using Triton X-100, the signaling of 1-pyrenylboronic acid was markedly enhanced. Herein, a spectrofluorimetric method for highly selective and sensitive hypochlorite determination has been performed. It can be noted that the fluorescence intensities positively correlated with the hypochlorite concentration over the range of 0.69–6.0 μmol L−1. The detection limit was 0.21 μmol L−1, which is lower than for most of the recently published methods. The experimental conditions were optimized and the effects of coexisting substances are evaluated. The results showed excellent priority because a certain amount of ions, including SO32−, NH4+, Cu2+ and other acid radicals, would not interfere with the measurement. The accuracy and reliability of the method was further ensured by recovery studies using the standard-addition method. In addition, the quenching mechanism, which was proven to be static quenching, has been investigated systematically by the linear plots at varying temperatures based on the Stern–Volmer equation, fluorescence lifetime, and UV-visible absorbance spectra. This method was finally used to detect hypochlorite in local water samples.

 Please wait while we load your content...
Please wait while we load your content...