Morphology and crystal structure control of α-Fe2O3 films by hydrothermal-electrochemical deposition in the presence of Ce3+ and/or acetate, F− ions
Abstract
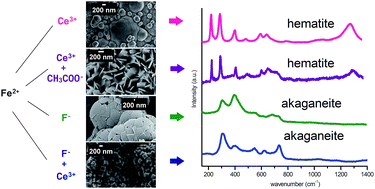
Hydrothermal-electrochemical growth of Hematite (α-Fe2O3) thin films in the presence of Ce3+ and/or CH3COO− and F− ions is reported. Primary attention is paid to understanding the synergistic effect of temperature and additive ions on the growth of Hematite particles. The literature describes the shape-controlled electrodeposition of iron oxide films, but these reports involve low-temperature depositions (<100 °C) that require a post-annealing step to obtain the Hematite phase. We studied size and shape control of Hematite crystals by introducing capping agents during hydrothermal-electrochemical deposition (HED) at 130 °C under autonomous pressure. When HED is used together with growth-modifying species such as CH3COO− and/or Ce3+, Hematite thin films with various structures including spherical, rhombohedral and platelet-like particles were obtained which were crystallized directly in the α-Fe2O3 phase without thermal annealing. In contrast, either Lepidocrocite or an amorphous phase forms with low-temperature electrodeposition under otherwise identical conditions. Addition of F− into the deposition solution induces formation of bundles that are composed of square planar Akaganeite particles even during HED. The aspect ratio of the Akaganeite particles is controlled with co-addition of Ce3+ ions. This variety of nanostructures gives us the opportunity to evaluate the photoelectrochemical performance of the α-Fe2O3 films based on particle size and shape. It was demonstrated that porous, square planar α-Fe2O3 with a high aspect ratio shows the highest performance.

 Please wait while we load your content...
Please wait while we load your content...