Dioxygen binding at a four-coordinate cobaltous porphyrin site in a metal–organic framework: structural, EPR, and O2 adsorption analysis†
Abstract
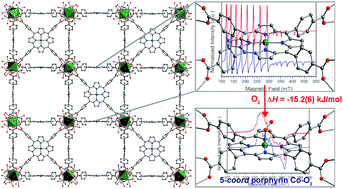
The study of cobalt porphyrin interactions with O2 is important owing largely to its relevance to biological heme-mediated O2 transport and storage. The immobilization of a metalloporphyrin site in a metal–organic framework (MOF) enables the study of these interactions without interference from solution effects such as bimolecular reactions, axial ligation, and solvent/solute interactions involving the porphyrin ligand. Here, we investigate the reaction of O2 with the four-coordinate cobaltous porphyrin complex in the MOF PCN-224Co. Single-crystal X-ray diffraction and electron paramagnetic resonance (EPR) spectroscopy of the oxygenated form, PCN-224CoO2 reveals a five-coordinate low-spin CoIII center coordinated to an S = 1/2 superoxo (O2−˙) ligand. Moreover, O2 adsorption measurements on PCN-224Co reveal two distinct binding sites with adsorption enthalpies of hads = −15.2(6) and −10.2(3) kJ mol−1. The former binding event is ascribed to ligation of O2 at the open Co site, and the obtained value is considerably lower than those observed for cobalt porphyrin units in substituted proteins and model complexes that feature axial ligands. These results provide the first structurally-characterized five-coordinate Co–O2 species, further highlight the importance of axial ligation in biological O2 transport and storage, and demonstrate the ability of a MOF to enable isolation and study of a species that is highly unstable in molecular form.
- This article is part of the themed collection: 2015 Emerging Investigators by ICF

 Please wait while we load your content...
Please wait while we load your content...