Nanocrystalline iron oxide based electroactive materials in lithium ion batteries: the critical role of crystallite size, morphology, and electrode heterostructure on battery relevant electrochemistry
Abstract
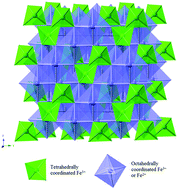
The importance of crystallite size control and direct synthesis of materials with desirable properties is broadly applicable for the rational design and development of new active materials for energy storage. Recently, the use of nanoparticles and crystallite size control has redefined electrode design strategies, due in part to the large surface area/volume ratios providing more pathways for ion movement within the bulk electrode. This review is structured primarily as a case study, where reports involving a specific densely structured iron oxide, magnetite, Fe3O4, and its use as an electrode in LIBs are used as examples. Due to the high theoretical capacity (924 mA h g−1), and opportunity for implementation of a low cost electrode material, magnetite was selected as the model material for this review. Notably, crystallite size, morphology, and electrode heterostructure can all play a critical role in battery relevant electrochemistry, particularly for crystallographically dense materials such as Fe3O4. Several examples of Fe3O4 based composites are described, incorporating different types of conductive materials such as carbons as part of the structure. Additionally, this review also provides a brief introduction to a newer iron oxide based material with a 2D layered structure, silver ferrite, where crystallite size control was synthetically achieved. By focusing on two specific iron oxide based nanoscale inorganic materials, this review highlights and distinguishes the contributions of electroactive material crystallite size, morphology and electrode heterostructure to electrochemical behavior, facilitating the future development of next generation of battery electrodes.
- This article is part of the themed collection: Inorganic Chemistry Advancing the Next Generation Batteries

 Please wait while we load your content...
Please wait while we load your content...