Synthesis and characterization of responsive poly(anionic liquid) microgels†
Abstract
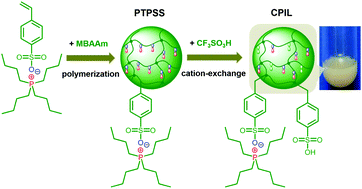
We report a class of poly(anionic liquid) microgels that undergo stimuli-responsive volume phase transitions. Such a microgel is synthesized by free radical precipitation polymerization of a tetrabutylphosphonium 4-styrenesulfonate monomer and a cross-linker N,N’-methylenebisacrylamide. These microgels can not only undergo reversible volume phase transitions in response to changes in temperature in water, methanol, or water/methanol mixtures, but also undergo re-entrant swelling–shrinking–swelling transitions as the methanol content increases in water/methanol mixtures at a set temperature in our experimental temperature window of 25.0–64.0 °C. Such microgels can be post functionalized, e.g., via ion-exchange treatment with HSO3CF3 to partially transform sulfonate to the catalytic Brønsted acidic –SO3H, whilst the yielded microgels can inherit the responsive properties. With both responsive and catalytic properties simultaneously harnessed on the same object, the functionalized microgels potentially can be used as a highly efficient catalyst for the esterification reaction of palmitic acid and the transesterification reaction of tripalmitin with methanol (as model reactions) at 65.0 °C (temperature of an oil bath), and allow the catalytic activity to be modulated to a certain extent in a non-monotonous way, making it possible to boost the reactions at a relatively lower temperature (e.g., 42.0 °C), while maintaining considerable catalytic activity. These features underlie the feasible use of the microgels in biodiesel production (with waste cooking oil as a model feedstock).

 Please wait while we load your content...
Please wait while we load your content...