Polymerization of trimethylene carbonates using organic phosphoric acids†
Abstract
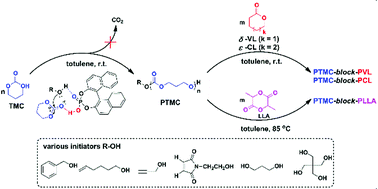
The homopolymerization of trimethylene carbonates (TMC) and their diblock copolymerization to PTMC-b-PVL and PTMC-b-PCL were carried out using benzyl alcohol (BnOH) as the initiator and 1,1′-binaphthyl-2,2′-diyl hydrogen phosphate (BNPH) as the catalyst in toluene at room temperature. The measured 1H NMR molecular weight (PTMC, Mn, NMR = 5300 g mol−1) and the theoretical one (PTMC, Mn, theo = 5100 g mol−1) calculated from the initial ratio of [TMC]0/[BnOH]0 and the monomer conversion matched ideally. The 1H NMR, SEC and MALDI-ToF MS measurements clarified that the obtained polymers were exactly as expected. Kinetics and chain extension experiments demonstrated the controlled/living nature for the ROP of TMC using BNPH. NMR titration experiments confirmed that the polymerization proceeded via a bifunctional activation mechanism. Extension of the initiators by using multifunctional 1,3-propanediol, pentaerythritol, propargyl alcohol, 5-hexen-1-ol, and N-(2-hydroxyethyl)maleimide installed α,ω-dihydroxy telechelic, star-shaped, and clickable end-functionalized polycarbonates. Block copolymers poly(trimethylene carbonate)-block-poly(δ-valerolactone), poly(trimethylene carbonate)-block-poly(ε-caprolactone), and poly(trimethylene carbonate)-block-poly(L-lactide) had been successfully obtained, which suggests that the BNPH catalysis is broadly applicable.


 Please wait while we load your content...
Please wait while we load your content...