Well-defined 4-arm stars with hydroxy-terminated polyethylene, polyethylene-b-polycaprolactone and polyethylene-b-(polymethyl methacrylate)2 arms†
Abstract
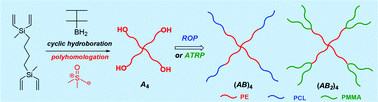
Bis-boron-thexyl-silaboracycle was prepared by hydroboration of 1,4-bis(methyldivinylsilyl)butane with thexylborane and used to initiate the polyhomologation of dimethylsulfoxonium methylide to afford well-defined hydroxy-terminated 4-arm polyethylene (PE) stars. The synthesized PE stars were transformed to (PE-b-PCL)4 starblock copolymers via the ring-opening polymerization of ε-caprolactone (CL) initiated by the hydroxyl end groups of (PE-OH)4 in the presence of P2-tBu phosphazene base. Esterification of the hydroxyl groups of the OH-terminated PE star with 2,2-dichloroacetyl chloride led to (PE-Cl2)4 which was used as initiator (eight initiating atom transfer radical polymerization, ATRP, sites) for the synthesis of (PE-b-PMMA2)4 dendrimer-like stars by the ATRP of methyl methacrylate (MMA). All intermediates and final products were characterized by high temperature gel permeation chromatography and proton nuclear magnetic resonance spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...