Synthesis of well-defined carboxyl poly(ε-caprolactone) by fine-tuning the protection group†
Abstract
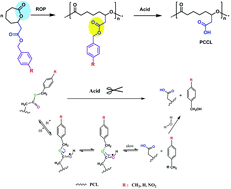
The introduction of reactive groups such as –NH2, –COOH etc. onto a poly(ε-caprolactone) (PCL) backbone was necessary for further modification but a well-controlled approach remains a challenge for synthetic chemistry. Carboxyl functionalized PCL was typically prepared via three steps involving the synthesis of the corresponding monomer with a carboxyl-protecting group, polymerization and the removal of the protection. Except for obtaining purified monomers and a decent polymerization, the most critical step in carboxyl PCL synthesis was the deprotection from the degradable main chain. Therefore, electronic effects and steric hindrance of the protecting group were taken into account with the aim for controllable polymerization and feasible deprotection. Substituents including –CH3, H and NO2 with discriminative electronegativity on the para position of the benzyl protecting group have been selected to investigate their behavior in monomer preparation, polymerization and deprotection, respectively. It turned out that the electron donating group (–CH3) displayed the highest selectivity in the monomer preparation, excellent control over the polymerization degree and the most efficient removal of the protecting groups without degradation of the backbone. In addition, the reactivity of the pendant carboxyl groups on PCL was demonstrated by amidation with 4-amino-2,2,6,6-tetramethylpiperidinyloxy (4-amino-TEMPO). Our results also provide guidance information on preparing well-defined biodegradable polymers with pendant reactive groups such as polypeptides, expanding the library of novel biomaterials.

 Please wait while we load your content...
Please wait while we load your content...