An amphiphilic block copolymer conjugated with carborane and a NIR fluorescent probe for potential imaging-guided BNCT therapy†
Abstract
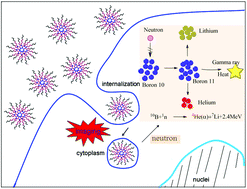
A carborane-containing triblock copolymer conjugated with a near infrared (NIR) fluorescence probe has been synthesized via reversible addition fragmentation chain transfer (RAFT), ring open polymerization (ROP), and conjugations of a cyanine NIR dye. Reactions were traced by means of 1H-NMR, GPC and FT-IR. The NIR amphiphilic copolymer with polycaprolactone and polycarborane as hydrophobic segments and poly(ethylene glycol)methyl ether methacrylate as the hydrophilic segment could self-assemble into micelles in aqueous solution, and was characterized by means of DLS and TEM. In the copolymer, carborane works as a boron neutron capture therapy (BNCT) agent while the aminocyanine (Cy) dye works as a NIR bioimaging agent, and the nanoparticles of the copolymers are a potential theranostic BNCT system for cancer therapy, which was supported by MTT experiments and in vitro fluorescence microscopy studies.


 Please wait while we load your content...
Please wait while we load your content...