Ring opening metathesis polymerization of cyclopentene using a ruthenium catalyst confined by a branched polymer architecture†
Abstract
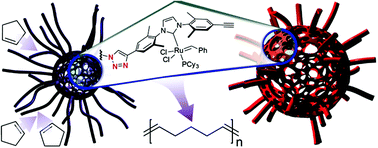
Multi-arm polystyrene stars functionalized with Grubbs-type catalysts in their cores were synthesized and used for the ring-opening metathesis polymerization (ROMP) of cyclopentene. The spatial confinement of the catalytic sites and the nanoscale phase separation between polystyrene and the growing polypentenamer chains lead to a dramatic inhibition of the ROMP termination and chain transfer steps. Consequently, cyclopentene polymerizations proceeded fast and with a high degree of conversion even in air. The Grubbs second generation catalyst was oxidatively inactivated under the same conditions. In contrast to conventional small-molecule catalysts, the ultimate degree of conversion of the cyclopentene monomer and the polydispersity of the product polypentenamer are not affected by the temperature. This indicates that spatial confinement of the catalyst results in a significant change in the activation parameters for the alkene metathesis ring-opening.

 Please wait while we load your content...
Please wait while we load your content...