Diblock metallocopolymers containing various iron sandwich complexes: living ROMP synthesis and selective reversible oxidation†
Abstract
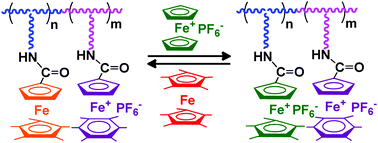
The design of redox-robust metallocopolymers is expected to produce new nanomaterials with multiple applications. Here the ROMP syntheses using Grubbs’ 3rd generation catalyst of three new living diblock copolymers each containing two distinct redox-stable iron sandwich complexes of the ferrocene, pentamethylferrocene and cyclopentadienyl-iron-arene families in the side chain are reported. The electrochemical properties of these diblock metallocopolymers investigated by cyclic voltammetry show complete chemical and electrochemical reversibilities of the redox waves, which also allow using the Bard–Anson method to determine the number of monomer units. The selective and reversible “chemical” oxidation of one of the blocks in two of these metallocopolymers afforded the syntheses of mixed-valent FeIIFeIII polymers.

 Please wait while we load your content...
Please wait while we load your content...