Studies on homologous random and alternating segmented conjugated polymers with and without silicon synthesized by ADMET†‡
Abstract
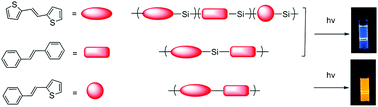
Using acyclic diene metathesis (ADMET), we synthesized homologous luminescent conjugated polymers with two aromatic segments based on thiophene and substituted phenylene, either alternating or randomly distributed, and either directly connected or separated by Si-linkers. In the polymers with Mn ∼ 5000–14 600 g mol−1, random or alternating placement of two segments yielded similar electro-optical properties (absorption λmax at ∼363 nm, emission, λmax ∼ 411 nm, HOMO–LUMO gap 3.00–3.07 eV), although alternating segments showed a slightly more defined absorption with slightly higher absorptivity and emission efficiency (57% vs. 51%). The alternating segments directly conjugated (without Si-linkers) showed a longer wavelength absorption and emission (λmax at ∼490 nm and ∼556 nm respectively), and slightly lower emission efficiencies (40%), as well as a smaller HOMO–LUMO gap of 2.21 eV. DFT calculations support the above results and analyses. Understanding such specific interactions between the aromatic units might provide guidance for future designs of segmented conjugated polymers.

 Please wait while we load your content...
Please wait while we load your content...