Chiral phosphoric acid-catalyzed asymmetric transfer hydrogenation of 3-trifluoromethylthioquinolines†
Abstract
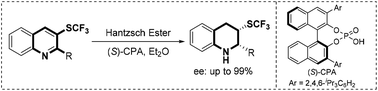
A chiral phosphoric acid-catalyzed asymmetric transfer hydrogenation of 3-trifluoromethylthioquinolines has been successfully developed, providing direct and facile access to chiral 2,3-disubstituted 1,2,3,4-tetrahydroquinoline derivatives containing a stereogenic trifluoromethylthio group with up to 99% enantioselectivity.

 Please wait while we load your content...
Please wait while we load your content...