Chiral phosphoric acid catalyzed asymmetric addition of naphthols to para-quinone methides†
Abstract
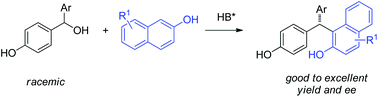
An asymmetric addition of naphthols to in situ generated para-quinone methides catalyzed by a chiral phosphoric acid is described. A range of useful triarylmethanes can be generated from stable general para-hydroxybenzyl alcohols with good efficiency and enantioselectivity.
- This article is part of the themed collection: New Talent

 Please wait while we load your content...
Please wait while we load your content...