Synthesis of α-diazo-β-keto esters, phosphonates and sulfones via acylbenzotriazole-mediated acylation of a diazomethyl anion†
Abstract
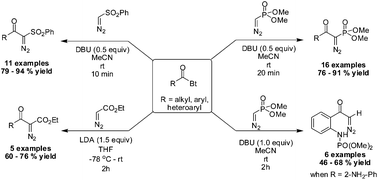
We report a method for the synthesis of α-diazo-β-keto esters, phosphonates and sulfones via acylation of corresponding diazomethyl anions with N-acylbenzotriazoles. The N-o-amino-acylbenzotriazoles exhibited an unprecedented transphosphorylation reaction leading to diazoacetyl phenylphosphoramidates.

 Please wait while we load your content...
Please wait while we load your content...