Multivalency effects on Pseudomonas aeruginosa biofilm inhibition and dispersal by glycopeptide dendrimers targeting lectin LecA†
Abstract
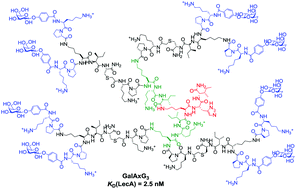
The galactose specific lectin LecA partly mediates the formation of antibiotic resistant biofilms by Pseudomonas aeruginosa, an opportunistic pathogen causing lethal airways infections in immunocompromised and cystic fibrosis patients, suggesting that preventing LecA binding to natural saccharides might provide new opportunities for treatment. Here 8-fold (G3) and 16-fold (G4) galactosylated analogs of GalAG2, a tetravalent G2 glycopeptide dendrimer LecA ligand and P. aeruginosa biofilm inhibitor, were obtained by convergent chloroacetyl thioether (ClAc) ligation between 4-fold or 8-fold chloroacetylated dendrimer cores and digalactosylated dendritic arms. Hemagglutination inhibition, isothermal titration calorimetry and biofilm inhibition assays showed that G3 dendrimers bind LecA slightly better than their parent G2 dendrimers and induce complete biofilm inhibition and dispersal of P. aeruginosa biofilms, while G4 dendrimers show reduced binding and no biofilm inhibition. A binding model accounting for the observed saturation of glycopeptide dendrimer galactosyl groups and LecA binding sites is proposed based on the crystal structure of a G3 dendrimer LecA complex.
- This article is part of the themed collection: Multivalent Biomolecular Recognition

 Please wait while we load your content...
Please wait while we load your content...